Summary: Inspire Medical Systems has received approval under the European Union Medical Device Regulation (EU MDR) for full-body MRI scans for patients using its Inspire therapy for obstructive sleep apnea. This approval, which expands the previous scan limitations, applies retroactively to all patients with the Inspire IV neurostimulator device.
Key Takeaways:
- Expanded MRI Compatibility: The EU MDR approval now allows Inspire therapy patients to undergo full-body MRI scans, broadening the scope from the previously permitted head, neck, and extremity scans.
- Retroactive Application: The new approval is retroactive, meaning it applies to all patients with the Inspire IV neurostimulator device, which was introduced in 2018.
- Enhanced Patient Care: This milestone addresses a significant barrier for patients concerned about MRI access, according to Inspire.
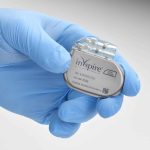
Inspire Medical Systems, a medical technology company focused on the development of minimally invasive solutions for patients with obstructive sleep apnea (OSA), announced that the European Union Medical Device Regulation (EU MDR) has approved additional magnetic resonance imaging (MRI) scan conditions for use with Inspire therapy.
This full-body MRI approval expands the Inspire use labeling that previously allowed only head, neck, and extremity MRI scans. Most importantly, this approval is retroactive, applying to all patients with the Inspire IV neurostimulator device, introduced in 2018, already in place.
“Expanding compatible use to include full-body MRI is a significant milestone in our effort to bring Inspire to more obstructive sleep apnea patients who struggle with CPAP. Providing the full range of scan options enables us to better help all current and future patients with their imaging needs,” says Tim Herbert, chairman and CEO of Inspire, in a release. “This full-body MRI compatibility has been an important benefit for patients in the United States for the past two years, and this new approval will provide the same positive benefit for patients in Europe going forward.”
MRI scanners use powerful magnets and radiofrequency energy to create detailed images of the inside of the body. Every year, millions of MRIs are performed in Europe to evaluate cancer, neurological, musculoskeletal, and other conditions. Inspire notes it has completed extensive testing to validate performance in the 1.5T MRI environment and demonstrate the conditions that allow scans to be performed safely.
“Until now, concern over future access to MRI had been a barrier for some patients considering Inspire therapy. Compatibility with this important diagnostic tool will provide peace of mind for current and future Inspire patients,” says Andreas Henke, executive vice president, managing director Europe, in a release.
Photo 86248177 © Tyler Olson | Dreamstime.com

Leave a Reply