Summary: Vyvo Smart Chain has received FDA 510(k) clearance for its wearable health and wellness tracking device, LifeWatch. This device promotes a user-centric health monitoring model and leverages Decentralized Physical Infrastructure network technology to enable users to … [Read more...]
DeltaWave Nasal Pillow CPAP Mask Receives FDA Clearance
Summary: REMSleep Holdings Inc has received FDA 510(K) clearance for its new DeltaWave nasal pillow CPAP interface mask. The DeltaWave is designed to enhance comfort and maintain natural breathing patterns, aiming to address common issues that lead to therapy discontinuation. The … [Read more...]
Genio OSA: Nyxoah Submits Final PMA Application to FDA
Summary: Nyxoah has submitted the fourth and final module of its Premarket Approval (PMA) application to the FDA for its Genio neurostimulator, designed to treat obstructive sleep apnea (OSA). The Genio system, which is fully-body MRI-compatible and powered by a wearable, … [Read more...]
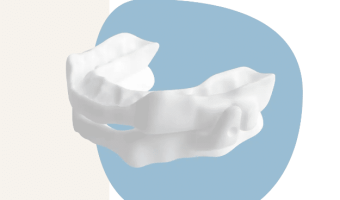
Australian-Designed Oral Appliance Gains FDA Clearance
Summary: GoodSleepCo, an Australian medical device company, has received FDA 510(k) clearance for its hushd Pro Avera oral appliance, which treats snoring and mild to moderate obstructive sleep apnea. Custom-made splints reposition the lower jaw to prevent airway blockage during … [Read more...]
FDA Rejects Petition Challenging Safety of Narcolepsy Drug Wakix
Summary: The FDA has denied a citizen petition filed by a short seller challenging the safety and effectiveness of Wakix (pitolisant) for treating excessive daytime sleepiness and cataplexy in adults with narcolepsy. The FDA reaffirmed Wakix’s favorable benefit-risk profile and … [Read more...]
FDA Approves Non-Scheduled Narcolepsy Drug for Children
Summary: The FDA has expanded the approval of a narcolepsy drug, previously approved for adults, to now include children aged 6 and older. This new approval provides a non-scheduled treatment option for pediatric patients experiencing excessive daytime sleepiness due to … [Read more...]
FDA Clears Snap’s Next-Gen Wearable Home Sleep Test
Summary: Snap Diagnostics has received FDA 510(k) clearance for its new wearable home sleep testing device, the Sleep Apnea Monitor (SAM) Model 9-10000. This next-generation device is designed to be smaller, easier to set up, and offers enhanced diagnostics compared to its … [Read more...]
FDA Clears EEG Brain Monitoring Headset
Summary: The US Food and Drug Administration has granted 510(k) clearance to Zeto Inc’s next-generation electroencephalography (EEG) brain monitoring device, ONE, for use in various settings including hospitals, homes, ambulances, and air transport. The device, which features 21 … [Read more...]
Nyxoah Prepares Final FDA Module for Genio OSA Neurostimulator
Summary: Nyxoah described its Q1 2024 economic and operating outcomes, highlighting sizeable progress with its Genio program, a leadless and battery-cost-free hypoglossal neurostimulation therapy for obstructive snooze apnea (OSA). The Aspiration US pivotal study reached co-key … [Read more...]
FDA Said It Never Inspected Dental Lab That Made Controversial AGGA Device
By Brett Kelman and Anna Werner, CBS News May 13, 2024 The FDA never inspected Johns Dental Laboratories during more than a decade in which it made the Anterior Growth Guidance Appliance, or “AGGA,” a dental device that has allegedly harmed patients and is now the subject of a … [Read more...]